Rift Valley Fever Virus
Rift Valley fever (RVF) is an acute disease of domestic ruminants in Africa and the Arabian Peninsula. This disease is caused by a mosquito-borne virus of the family Bunyaviridae and genus Phlebovirus. Large outbreaks occur at irregular intervals when heavy rains favor breeding of mosquito vectors of the virus and are characterized by deaths of newborn animals and abortion in pregnant sheep, goats, and cattle. Recent outbreaks outside Africa have led to rediscover the human disease but it remains poorly known. The wide spectrum of acute and delayed manifestations with potential unfavorable outcome complicate the management of suspected cases and prediction of morbidity and mortality during an outbreak. Although RVF often causes severe disease in animals, most people with RVF have either no symptoms or a mild illness with fever, weakness, back pain, and dizziness. However, a small percentage of about 10% of people with RVF develop much more severe symptoms, including hemorrhage, eye disease, and encephalitis.
Discover our Products for Research on WHO Priority Diseases!
Zikavirus, SARS-CoV-2, SARS, Ebolavirus, Marburgvirus, RVFV & more
Areas with the most recent RVF outbreaks include Somalia (2006-7), Kenya (2006-7), Tanzania (2007), Sudan (2007-8), Mayotte (2007-8), Madagascar (2008), Swaziland (2008) and South Africa (2008, 2009, and 2010). In affected areas, RVFV epizootics cause catastrophic livestock losses. From November 2018 through July 2019, an outbreak of Rift Valley fever in humans occurred in Mayotte, France; 142 cases were confirmed. Exposure to animals or their biological fluid was reported by 73% of patients. As other arboviral infections including dengue, chikungunya and zika, RVF is emerging worldwide, due to the globalization of arthropod vectors, mainly mosquitoes, which efficiently transmit an increasing number of old, unrecognized and new viruses. Arboviruses pose a major threat of introduction to several continents, including Europe and North America, with the possibility of co-circulation.
At present, Rift Valley Fever is one of the WHO priority diseases. Public health and animal health agencies agree that it is now a priority to develop RVFV vaccines (whether for humans, animals, or both) that will yield highly effective, long-term protective immunity. Past experience with killed inactivated RVFV vaccines has shown them to be protective but expensive to produce and inconvenient to administer, often requiring multiple booster immunizations to achieve and maintain protection.
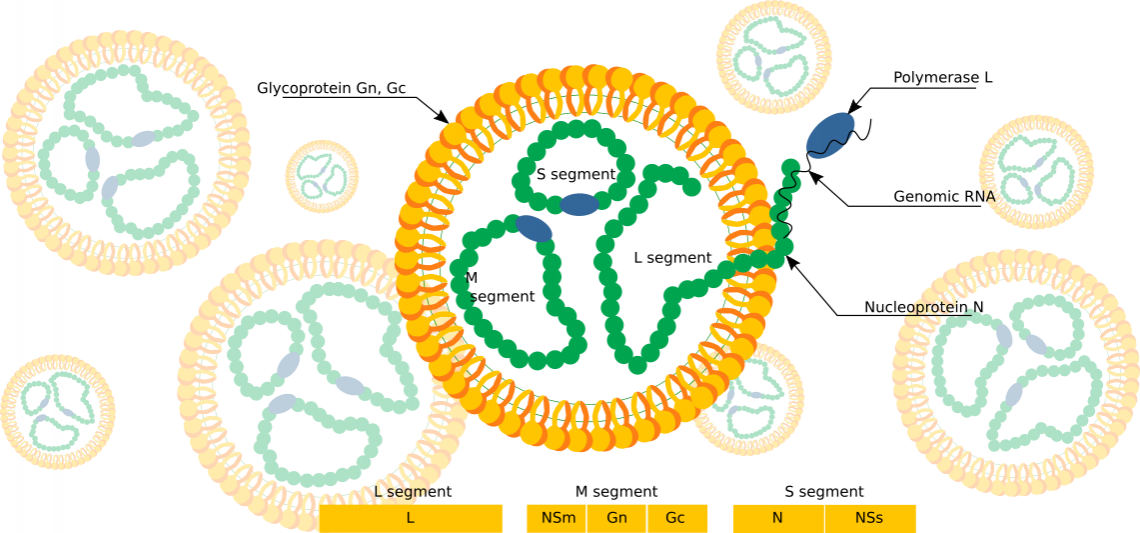
Fig. 1: Rift Valley Fever Virus Schema (6)(7)
Rift Valley Fever: Structure and Genome Organization
Rift Valley fever virus (RVFV) is one of the most important members of the large and diverse Bunyaviridae family. RVFV belongs to the Phlebovirus genus, which contains 9 other viral species including Punta Toro, Sandfly fever, and severe fever with thrombocytopenia syndrome (SFTS) virus. The name phleobovirus is derived from the fact that most of the viruses in this genus are transmitted by phlebotomine sandflies. The genome segments of bunyaviruses encode four structural proteins: the viral polymerase (L) on the large (L) segment, two glycoproteins (Gn and Gc) on the medium (M) segment, and the viral nucleocapsid protein (N) on the smallest (S) segment. RVFV additionally expresses two nonstructural proteins encoded on the M segment, a 78kDa (NSm1) protein and a 14kDa (NSm2) protein, and one on the S segment (termed NSs).
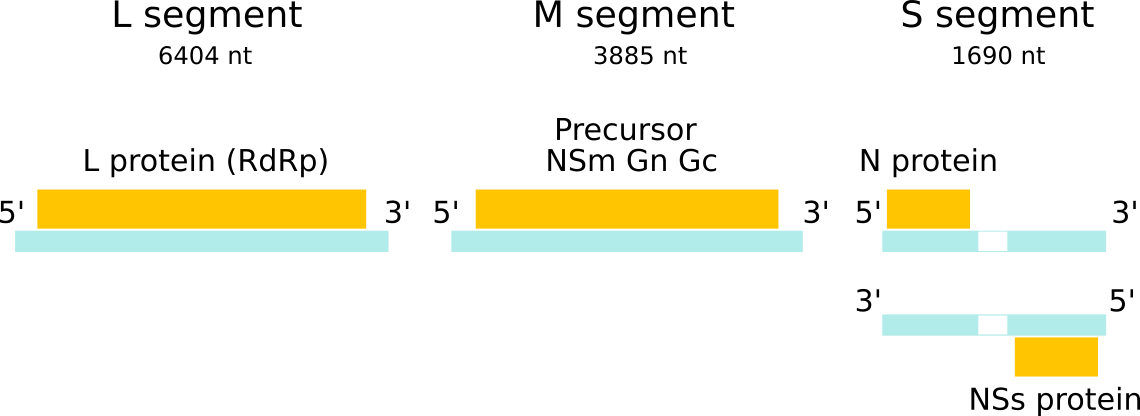
Fig. 2: The three genomic RNA segments of bunyaviruses contain untranslated regions (UTRs) at the 3’ and 5’ ends which are segment-specific and serve as promoters for transcription and replication by the viral polymerase. For RVFV, the L and M segments are of negative polarity while the S segment utilizes an ambisense strategy to code for both N and NSs.
Antibodies for Rift Valley Fever Virus Research
antibodies-online offers the following research antibodies for laboratory research on RVFV. For more details click on the product links below. In case of any question do not hesitate to contact our scientific customer support.
Recombinant Proteins for Rift Valley Fever Virus Research
antibodies-online offers you recombinant His-tagged RVFV L, NS, NP proteins for laboratory research. For more details click on the product links below. In case of any question do not hesitate to contact our scientific customer support.
Viral Peptides
Below you will find a selection of viral peptides for blocking the activity of RVF virus antibody.
References
: "Rift valley fever: recent insights into pathogenesis and prevention." in: Journal of virology, Vol. 85, Issue 13, pp. 6098-105, (2011) (PubMed).: "Virulence factor NSs of rift valley fever virus recruits the F-box protein FBXO3 to degrade subunit p62 of general transcription factor TFIIH." in: Journal of virology, Vol. 88, Issue 6, pp. 3464-73, (2014) (PubMed).
: "Rift Valley Fever Outbreak, Mayotte, France, 2018-2019." in: Emerging infectious diseases, Vol. 26, Issue 4, pp. 769-772, (2021) (PubMed).
: "Advances in Rift Valley fever research: insights for disease prevention." in: Current opinion in infectious diseases, Vol. 23, Issue 5, pp. 403-8, (2010) (PubMed).
: "The challenging management of Rift Valley Fever in humans: literature review of the clinical disease and algorithm proposal." in: Annals of clinical microbiology and antimicrobials, Vol. 19, Issue 1, pp. 4, (2020) (PubMed).
: "Molecular epidemiology of Rift Valley fever virus." in: Emerging infectious diseases, Vol. 17, Issue 12, pp. 2270-6, (2012) (PubMed).

Master of Science in engineering. 12+ years of experience in marketing and e-commerce in the life science sector.
Go to author page



