Haemophilus Influenzae antibody
-
- Target
- Haemophilus Influenzae
- Reactivity
- Haemophilus influenzae
-
Host
- Mouse
-
Clonality
- Monoclonal
-
Conjugate
- Un-conjugated
- Application
- Enzyme Immunoassay (EIA), Immunofluorescence (IF)
- Purification
- Affinity Chromatography on Protein G
- Immunogen
- Native, NCTC 7279.
- Clone
- 1079-83
- Isotype
- IgG2b
-
-
- Application Notes
-
ELISA. Immunofluorescence.
Other applications not tested.
Optimal dilutions are dependent on conditions and should be determined by the user. - Restrictions
- For Research Use only
-
- by
- Department of Medical Biochemistry and Cell Biology, University of Gothenburg
- No.
- #101185
- Date
- 08/31/2017
- Antigen
- H. influenzae
- Lot Number
- 1601
- Method validated
- Enzyme Immunoassay
- Positive Control
- Purified human lung mucin incubated with Haemophilus influenzae and ABIN180862
- Negative Control
- Purified human lung mucin incubated with ABIN180862 without Haemophilus influenzae
- Notes
- Passed, the antigen Haemophilus influenzae antibody ABIN180862 specifically detects its antigen in human mucin samples incubated with the bacteria.
- Primary Antibody
- ABIN180862
- Secondary Antibody
- anti-mouse IgG antibody HRP conjugate (Merck, AP160P)
- Full Protocol
- Human lung mucin isolation and purification
- Isolate mucins from bronchoalveolar lavage fluid of 8 human patients.
- Place samples in five volumes of extraction buffer (6M guanidinium chloride (GuHCl), 5mM EDTA, 10mM sodium phosphate buffer pH6.5) containing 0.1 M PMSF.
- Stir samples slowly ON at 4°C.
- Reduce samples with 10ml of 10mM 1,4-dithiothreitol (DTT) in reduction buffer (6M guanidinium chloride, 5mM EDTA, 0.1M Tris-HCl buffer pH8.0) for 5h at 37°C.
- Alkylate samples with 25mM iodoacetamide in the dark ON at 23-24°C.
- Centrifuge samples at 23000xg for 50min at 4°C in a Beckman JA-30 rotor.
- Dialyze samples against 10 volumes of extraction buffer for 24h at 4°C.
- Adjust sample volume to 26ml with extraction buffer.
- Add CsCl to a starting density of 1.39g/ml by gentle stirring.
- Transfer samples to Quick Seal ultracentrifuge tubes (Beckman, 342699).
- Centrifuge samples at 40000xg for 90h at 15°C in a Beckman 70-Ti rotor.
- Collect fractions from the bottom of the tubes.
- Glycan detection
- Dilute gradient fractions in 0.5M GuHCl.
- Coat fractions onto 96-well PolySorp plates (ThermoFisher Scientific, 475094) ON at 4°C.
- Wash plates 3x with DELFIA washing solution (5mM Tris-HCl, 0.15M NaCl, 0.005% Tween 20, 0.01% NaN3, pH7.75).
- Periodate oxidation of polysaccharide conjugates with 25mM sodium metaperiodate in 0.1M sodium acetate buffer pH5.5 for 20min at 22-24°.
- Wash plates 3x with 200µl/well DELFIA washing solution.
- Wash plates once with 200µl/well PBS 0.05% Tween.
- Block plates with 200µl/well DELFIA blocking solution (50mM Tris-HCl, 0.15M NaCl, 90µM CaCl2, 4µM EDTA, 0.02% NaN3, and 0.1% BSA, 200µl/well) for 1h at 22-24°C.
- Discard blocking buffer.
- Incubated plates with 100µl/well 2.5µM biotin-hydrazide solution in 0.1M sodium acetate buffer pH5.5 for 1h at 22-24°C.
- Wash plates 3x with 200µl/well DELFIA washing solution.
- Incubate plates with 100µl europium labelled Streptavidin (PerkinElmer) diluted 1:1000 in DELFIA assay buffer (PerkinElmer) for 1h at 22-24°C.
- Wash plates 6x with 200µl/well DELFIA washing solution.
- Incubate plates with 200µl/well DELFIA enhancement buffer (PerkinElmer) on a shaker for 5min at 22-24°C.
- Measure fluorescence using a Wallac 1420 VICTOR2 plate reader following the europium label protocol (PerkinElmer, Waltham, MA, USA).
- Haemophilus influenzae binding to human lung mucins
- Coat mucin samples onto 96-well PolySorp plates (ThermoFisher Scientific, 475094) ON at 4°C.
- Wash plates 3x with washing buffer (PBS with 0.05% Tween 20).
- Block wells with 200µl/well 1% Blocking Reagent for ELISA (Roche) for 1h at 22-24°C.
- Discard blocking buffer.
- Dilute bacteria to an OD600 of 0.05 in blocking buffer containing 0.05% Tween 20.
- Add 100µl/well of the bacterial suspension to the coated plates.
- Incubate plates for 2h at 37°C at 120rpm on an orbital shaker.
- Wash plates 3x with 200µl/well washing buffer.
- Incubate plates with 100µl/well H. influenzae antibody (antibodies-online, ABIN180862, lot 1601) diluted 1:500 in blocking buffer for 1h at 22-24°C.
- Wash plates 3x with 200µl/well washing buffer.
- Incubate plates with 100µl anti-mouse IgG antibody HRP conjugate (Merck, AP160P) diluted 1:5000 in blocking buffer for 1h at 22-24°C.
- Wash plates 200µl/well washing buffer.
- Add 100µl/well TMB substrate to the wells.
- Incubate plates for 20min at 22-24°C.
- Stop reaction with an equivalent amount of 0.5 M H2SO4.
- Measure absorbance in a microplate reader at 450nm.
- Experimental Notes
Validation #101185 (Enzyme Immunoassay)![Successfully validated 'Independent Validation' Badge]()
![Successfully validated 'Independent Validation' Badge]() Validation Images
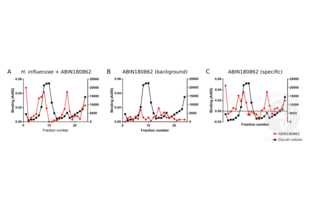
Validation Images![Binding of H. influenzae to human lung mucins. A. Bound H. influenzae is detected by ABIN180862. The antibody curve (red line) coincides with the mucin containing fractions (black line). B. Background signal of ABIN180862 antibody (i.e. antibody binding to mucin in the absence of H. influenzae). C. Binding of H. influenzae to human lung mucins after subtracting unspecific background signal.]() Binding of H. influenzae to human lung mucins. A. Bound H. influenzae is detected by ABIN180862. The antibody curve (red line) coincides with the mucin containing fractions (black line). B. Background signal of ABIN180862 antibody (i.e. antibody binding to mucin in the absence of H. influenzae). C. Binding of H. influenzae to human lung mucins after subtracting unspecific background signal.
Full Methods
Binding of H. influenzae to human lung mucins. A. Bound H. influenzae is detected by ABIN180862. The antibody curve (red line) coincides with the mucin containing fractions (black line). B. Background signal of ABIN180862 antibody (i.e. antibody binding to mucin in the absence of H. influenzae). C. Binding of H. influenzae to human lung mucins after subtracting unspecific background signal.
Full Methods -
- Concentration
- 1.0 mg/mL
- Buffer
- PBS, 0.09 % Sodium Azide
- Preservative
- Sodium azide
- Precaution of Use
- This product contains sodium azide: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
- Storage
- 4 °C/-20 °C
- Storage Comment
-
Store the antibody undiluted at 2-8 °C for one month or (in aliquots) at -20 °C for longer. Avoid repeated freezing and thawing.
Shelf life: one year from despatch. - Expiry Date
- 12 months
-
- Target
- Haemophilus Influenzae
- Target Type
- Species
-

 (1 validation)
(1 validation)



